Promising COVID-19 Vaccine Trial on Hold – Setback or Standard Procedure?
News Staff via CBN News – There’s been a potential setback in efforts to pursue a COVID-19 vaccine as one of the most promising trials has now been placed on hold.
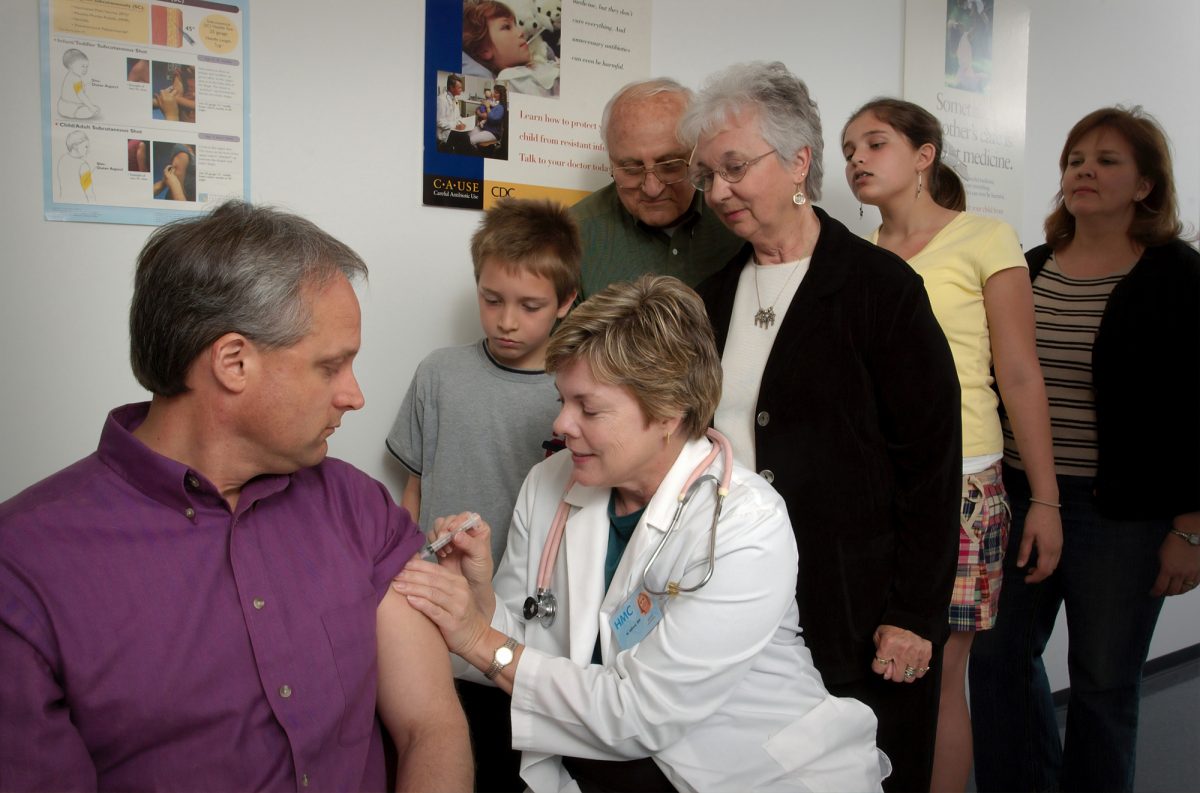
Researchers are pausing the stage three vaccine trial being conducted by Oxford University in the UK after a volunteer came down with an unexplained illness.
AstraZeneca, the company developing that vaccine with Oxford, is conducting an investigation to determine if the illness is linked to the trial.
In a statement, the company insisted that they won’t seek approval or emergency use until “demonstrating safety and efficacy through a phase 3 clinical study.
“The safety and efficacy of vaccines, including any potential vaccine for COVID-19, is reviewed and determined by expert regulatory agencies around the world, such as the United States Food and Drug Administration (FDA)” the statement reads. “FDA has established clear guidance for the development of COVID-19 vaccines and clear criteria for their potential authorization or approval in the US.”
Chief executives of nine drug companies, including AstraZeneca, BioNTech, Moderna, Pfizer, Novavax, Sanofi, GlaxoSmithKline, Johnson & Johnson, and Merck vowed not to rush a vaccine.
The pledge is intended to “help ensure public confidence in the rigorous scientific and regulatory process by which COVID-19 vaccines are evaluated and may ultimately be approved.”
Scientists are evaluating the public health impact of rushing a vaccine and the potential safety concerns, against the effect of delaying this highly anticipated treatment.
But, they point out that temporary halts are common in vaccine trials.
Meanwhile, two other vaccines are in stage-three trials in the US.
Just this week, President Trump said there could be a vaccine before Election Day. “So, we’re going to have a vaccine very soon. Maybe even before a very special date,” he stated.
To read the original article click here.
For more articles from CBN News click here.